RADO



Robotic Foot and Knee Orthosis (RADO)
Overview
The Robotic Foot and Knee Orthosis (RADO) is an innovative medical device in the microprocessor-controlled KAFO (knee-ankle-foot orthosis) category. By controlling the stance and swing phases of gait in real-time, it improves the lives of patients unable to walk safely due to quadriceps muscle insufficiency. It is designed for adults and adolescents with quadriceps paralysis or paresis, offering a wide range of applications from endoscopic to open surgeries, enabling independent living with a gait close to natural walking. RADO surpasses the limitations of traditional orthoses by providing a safer, more efficient, and comfortable solution with AI-based adaptation and a hydraulic-spring hybrid system.
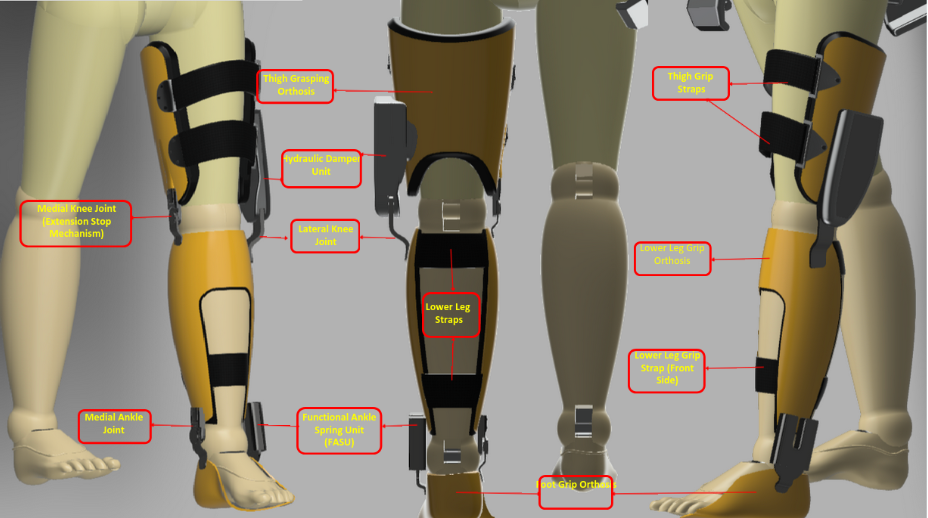

Technical Features and Integration
RADO is designed with a sensor fusion and microprocessor-controlled system:
● Sensor System: IMU (knee joint angle and acceleration), plantar pressure sensors (load transfer and phase detection), and armband EMG sensor (muscle activity measurement). Data is transmitted to the microprocessor for real-time analysis.
● Microprocessor Control: Operates with sensor fusion to distinguish stance/swing phases, locking the knee in milliseconds during potential fall scenarios.
● Hydraulic Damper: A hydraulic + spring hybrid mechanism provides resistance to flexion in the stance phase and allows controlled opening in the swing phase. The modular design ensures easy maintenance and repair.
● Software and AI: Machine learning-based user-specific adaptation. The "RADO" mobile app calibrates hydraulic resistance individually; automatic learning and personalization are completed within 14 days.
● Performance: 130 kg load capacity, total weight <2.9 kg, response time ≤0.20 ms, battery life 48 hours (80% charge in 2 hours). The system is integrable with existing KAFOs and aims for a 60% local contribution rate through domestic production.
Usage Areas and Disease Diagnoses
RADO is recommended for the following conditions:
● Incomplete Paraplegia: Reduced knee extension strength after L1-L5 spinal cord injury.
● Post-Polio Syndrome: Progressive quadriceps insufficiency following poliomyelitis.
● Peripheral Nerve Injuries: Femoral or sciatic nerve damage.
● Muscle Diseases: Types of muscular dystrophy, for preserving knee extension.
● Post-Traumatic Conditions: Weakness after lower extremity fractures or muscle/nerve cuts.
● Neurological Diseases: Multiple sclerosis (selected cases), peripheral neuropathies, upper motor neuron lesions. These solutions reduce fall risk, approximate natural gait patterns, and lower energy consumption by 30-45%.
Ethical and Clinical Validation
RADO is developed under ISO 13485 quality system standards, supported by clinical evaluation and post-market surveillance plans. Pilot studies will measure fall rate (targeting 80% reduction), gait tests (10MWT, TUG, 6MWT), balance (ABC scale), and quality of life (VAS, EQ-5D/SF-36). Field failure rate is targeted at ≤2%, with user satisfaction (QUEST 2.0, SUS ≥80/100) and donning/doffing time (≤90 seconds) assessed. The project prepares for CE/UKCA certification in compliance with AB MDR and UKCA regulations.
Benefits
● Safety and Precision: Minimizes fall risk with real-time phase control, reducing error rates.
● Flexibility and Comfort: Offers a near-natural walking experience, suitable for stairs and slopes; reduces fatigue by lowering energy consumption.
● Rapid Decision-Making: Ensures user safety with millisecond-level response times.
● Broad Application Area: Versatile for neurological, traumatic, and muscle-related conditions; enhanced accessibility with a local maintenance network.
Contact
For more information about the Robotic Foot and Knee Orthosis (RADO), to participate in beta testing, or to request a demo, please contact us.

